
TIG (Tungsten Inert Gas) welding has emerged as the gold standard for medical applications precisely because it delivers what healthcare demands: contamination-free joints, precise heat control that prevents material distortion, and welds smooth enough to withstand thousands of sterilization cycles. According to FDA's Quality Management System Regulation, effective February 2026, medical device welding now requires validated processes with complete traceability—making TIG's clean, documentable process more critical than ever.
This guide covers the essential applications of medical TIG welding, from surgical instruments to implantable devices, the materials and regulatory standards governing this specialized field, and the best practices that separate medical-grade welding from general industrial work.
TLDR: Key Takeaways
- TIG welding produces contamination-free welds essential for biocompatibility and patient safety
- Applications span surgical instruments, implants, medical gas systems, and pharmaceutical equipment
- Validated processes with full documentation meet FDA QMSR and ISO standards requirements
- Medical-grade materials like 316L stainless steel and titanium demand specialized welding parameters
- High-purity argon (≥99.999%) and cleanroom protocols distinguish medical welding
What is Medical TIG Welding?
TIG welding, also known as Gas Tungsten Arc Welding (GTAW), uses a non-consumable tungsten electrode to create an arc that melts the base metal. An inert shielding gas—typically argon—protects the weld pool from atmospheric contamination.
The welder can precisely control heat input by adjusting amperage, making TIG ideal for thin-walled medical components where distortion must be minimized.
What Makes TIG Welding "Medical-Grade"
Medical-grade TIG welding differs significantly from industrial applications through three critical requirements:
- Process Validation: The FDA classifies welding as a "special process" where results cannot be fully verified by inspection alone. Manufacturers must validate welding procedures before production and maintain documented proof that the process consistently produces acceptable welds.
- Contamination Control: Medical welding often occurs in Class 100 cleanrooms where particulate counts are strictly controlled. Even microscopic contamination can compromise biocompatibility or create sites for bacterial colonization.
- Complete Traceability: Every weld must be traceable to specific material lots, welder qualifications, and process parameters. For implantable devices, this documentation trail is mandatory under ISO standards for medical devices.

TIG vs. Other Medical Welding Methods
| Method | Best For | Limitations |
|---|---|---|
| TIG (GTAW) | Surgical instruments, sanitary piping, structural joints | Higher heat input than laser/electron beam |
| Laser Welding | Stents, pacemaker seals, micro-components | Equipment cost, limited penetration depth |
| Electron Beam | Implant assemblies, dissimilar metals | Requires vacuum chamber, high capital cost |
| Resistance Welding | Catheters, battery tabs, wire assemblies | Limited to spot/seam welds, electrode wear |
Among these methods, TIG excels where precision heat control meets structural integrity requirements—think reusable surgical forceps that must withstand thousands of autoclave cycles, or pharmaceutical tanks requiring smooth, crevice-free interior surfaces.
Manual vs. Automated TIG Welding
Manual TIG allows skilled welders to navigate complex geometries, repair damaged components, and adjust parameters in real-time. It's essential for prototype development and low-volume specialty devices.
Automated orbital TIG delivers repeatability for production runs. Orbital systems weld tubing and pipe connections with consistent bead profiles—critical for medical gas distribution systems where every joint must meet identical quality standards.
Documentation Requirements
Medical TIG welding requires documentation that general industrial work does not:
- Welding Procedure Specifications (WPS) validated through testing
- Welder Performance Qualifications (WPQ) requiring periodic recertification
- Material certifications that trace alloy chemistry to specific heats
- Process parameter records captured for every production weld
- Inspection reports with acceptance criteria based on ASME BPE or AWS D18.1 standards
Why TIG Welding is Essential for Medical Applications
Superior Weld Purity
The non-consumable tungsten electrode and inert gas shielding prevent the contamination that plagues other welding methods. There's no flux residue to trap bacteria, no spatter to create surface irregularities, and no consumable wire introducing unknown elements into the weld pool.
For implantable devices, this purity directly impacts biocompatibility—research shows that even minor contamination can trigger adverse tissue reactions or accelerate corrosion.
Precision Heat Control
TIG's adjustable amperage allows welders to precisely control heat input, preventing distortion in thin-walled components like catheter tips or delicate sensor housings. This control minimises the Heat Affected Zone (HAZ), preserving the base material's mechanical properties and corrosion resistance. For titanium implants, excessive heat causes grain coarsening that reduces ductility—TIG's precise control prevents this degradation.
Aesthetic Quality and Cleanability
Smooth, uniform weld beads aren't just cosmetic in medical devices—they're functional requirements. Rough welds create crevices where bacteria grow and cleaning solutions become trapped.
According to ASME BPE standards, pharmaceutical equipment welds must have specific surface finish requirements to enable effective cleaning and sterilisation. TIG naturally produces smooth beads with minimal post-weld finishing.
Material Versatility
TIG successfully welds the biocompatible alloys medical devices demand:
- Stainless steel 316L for surgical instruments and implant components
- Titanium Grade 5 and Grade 23 for orthopaedic implants
- Commercially pure titanium where corrosion resistance is paramount
- Nitinol for stents and shape-memory applications (with specialised parameters)

Regulatory Compliance
TIG welding's clean process and inherent documentation capabilities align perfectly with FDA and ISO standards requirements. The visible weld pool allows real-time monitoring, automated systems generate parameter logs automatically, and the process produces minimal defects requiring rework—all factors that simplify validation and ongoing quality assurance.
Key Medical Applications of TIG Welding
Surgical Instruments and Tools
TIG welding fabricates the forceps, scissors, retractors, and specialized tools that surgeons rely on daily.
These instruments face extreme demands: thousands of sterilization cycles without corroding, precise tolerances despite thermal cycling, and repeated mechanical stress.
Requirements include:
- Corrosion-resistant materials (typically 316L or 420 stainless steel)
- Smooth weld profiles that won't snag tissue or surgical gloves
- Joints strong enough to withstand sterilization thermal shock
- Dimensional stability through repeated autoclave cycles
Medical Implants and Prosthetics
Beyond surgical tools, TIG welding plays a critical role in implantable devices. Orthopedic implants like hip stems, trauma plates, and spinal fusion devices require TIG welding for structural subassemblies.
While laser welding dominates micro-components like cardiovascular stents, TIG handles larger structural joints where strength and fatigue resistance are critical.
Biocompatibility is non-negotiable. Any contamination in the weld can cause tissue inflammation or implant rejection. Materials must comply with ASTM F138 (316L stainless steel) or ASTM F136 (Ti-6Al-4V ELI) for implantable devices, and welding procedures must preserve these materials' corrosion resistance.

Diagnostic and Monitoring Equipment
Medical imaging equipment—MRI machines, CT scanners, ultrasound devices—contains precision-welded housings and sensor assemblies. These welds must provide electromagnetic shielding, maintain airtight seals protecting sensitive electronics, and withstand the vibration and thermal cycling of continuous operation.
Implantable diagnostic devices like neurostimulators require hermetic sealing tested to MIL-STD-883 standards, ensuring no moisture penetration over decades of implantation.
Medical Gas Delivery Systems
Hospital oxygen, nitrogen, and anesthetic gas systems demand leak-proof, contamination-free connections. ISO 7396-1:2016 specifies requirements for medical gas pipeline systems, including pressure testing to ensure systems contain only the intended gas.
TIG orbital welding creates consistent, full-penetration joints in stainless steel tubing that meet these requirements.
Every weld must be documented and inspected—patient safety depends on zero tolerance for leaks or cross-contamination between gas lines.
Pharmaceutical Processing Equipment
Bioreactors, mixing vessels, and transfer piping in pharmaceutical manufacturing require sanitary welding that prevents bacterial growth and contamination. ASME BPE governs these applications, specifying orbital GTAW for 304L and 316L stainless steel tubing.
Key requirements include:
- Smooth interior surfaces with no crevices (weld penetration must be complete)
- Minimal heat tint or oxidation that could harbor bacteria
- Full traceability of welding parameters and material certifications
- Visual acceptance criteria defined in ASME BPE Table MJ-8.4-1
Laboratory and Research Equipment
Analytical instruments, sample handling systems, and research apparatus often require custom fabrication with precise tolerances. TIG welding provides the chemical resistance needed for exposure to aggressive reagents while maintaining the dimensional accuracy research applications demand.
Materials Commonly Used in Medical TIG Welding
Stainless Steel 316L and 304
316L (Low Carbon) is the workhorse of medical device manufacturing. Defined by ASTM F138 for implants and ASTM F139 for sheet/strip, it offers excellent corrosion resistance and biocompatibility.
The "L" designation indicates low carbon content (≤0.03%), preventing carbide precipitation during welding that would compromise corrosion resistance.
304 Stainless Steel appears in non-implant applications like equipment housings and pharmaceutical processing equipment exteriors. It's more cost-effective than 316L but offers slightly lower corrosion resistance.
Both grades require careful welding technique:
Welding Considerations:
- ER308L or ER316L filler metals matching base metal
- Interpass temperature below 150°C prevents sensitization
- High-purity argon back purge prevents root oxidation
- Post-weld passivation per ASTM A967 restores oxide layer

Titanium and Titanium Alloys
Grade 5 (Ti-6Al-4V) offers high strength-to-weight ratio for orthopedic implants and surgical instruments.
Grade 23 (Ti-6Al-4V ELI) is the "Extra Low Interstitial" version with tightly controlled oxygen and iron content, providing superior ductility and fracture toughness for implants.
Commercially Pure Titanium (Grades 1-4) defined by ASTM F67 is used where maximum corrosion resistance matters more than strength—think dental implants or chemical processing equipment.
Critical Welding Requirements:
- Oxygen contamination above 10 ppm causes embrittlement
- Back purging is mandatory—both weld root and HAZ must be shielded
- High-purity argon (≥99.999%) for both torch and trailing shields
- Weld in controlled environments to prevent atmospheric contamination
- Any heat tint indicates oxygen pickup and requires removal
Nitinol (Nickel-Titanium)
ASTM F2063 covers Nitinol shape-memory alloys used in stents and medical springs. Welding Nitinol presents unique challenges—heat alters its transformation temperatures and superelastic properties. Oxide formation releases nickel ions that compromise biocompatibility.
Most Nitinol joining uses laser welding for minimal heat input, but TIG can work for larger components with carefully controlled parameters and immediate post-weld cleaning to remove oxides.
Quality Standards and Regulatory Compliance for Medical Welding
FDA Regulations
21 CFR Part 820 (Quality System Regulation) was harmonised with ISO standards on 2 February 2026. Key requirements affecting welding include:
- Process Validation (820.75): Welding must be validated with high assurance since results cannot be fully verified by inspection. This requires Installation Qualification (IQ), Operational Qualification (OQ), and Performance Qualification (PQ) protocols
- Production and Process Controls (820.70): Manufacturers must monitor welding parameters, maintain equipment, and document deviations from established procedures
ISO Standards Requirements
Clause 7.5.6 (Validation of processes) explicitly requires validation for special processes like welding. This includes qualifying equipment, procedures, and personnel before production begins.
Clause 7.5.9 (Traceability) requires systems to trace materials and components throughout manufacturing. For implantable devices, every weld must be traceable to specific material heats and welder qualifications.
Material Traceability
Medical device manufacturers must maintain complete material documentation:
- Mill test reports certifying alloy chemistry
- Heat numbers traceable to specific production lots
- Mechanical property test results
- Biocompatibility certifications where applicable
Together, this documentation proves that materials meet specifications like ASTM F138 or ASTM F136 and allows rapid recall if defects are discovered.
Weld Inspection and Testing
Visual Examination: ASME BPE Table MJ-8.4-1 defines acceptance criteria for weld bead width, uniformity, and profile. Discoloration levels are specified in Tables MJ-8.4-2 and MJ-8.4-3 based on whether tubing is electropolished or mechanically polished.
Dye Penetrant Testing (ASTM E165): Detects surface-breaking defects like cracks or incomplete fusion. Essential for welds in high-stress applications.
Radiographic Testing: X-ray examination reveals internal defects like porosity or lack of penetration that visual inspection cannot detect.
Destructive Testing: Qualification welds undergo tensile testing, bend testing, and metallographic examination to verify that procedures produce acceptable results before production begins.
Best Practices for Medical TIG Welding
Cleanliness Protocols
Pre-weld cleaning removes oils, oxides, and contaminants that would compromise weld quality:
- Degrease with non-chlorinated solvents or alkaline cleaners
- Remove oxides mechanically (stainless steel wire brush dedicated to the specific alloy) or chemically (pickling for stainless steel, etching for titanium)
- Final rinse with deionized water (18 MΩ quality for critical applications)
- Dry in clean, lint-free environment
- Handle with powder-free gloves to prevent recontamination
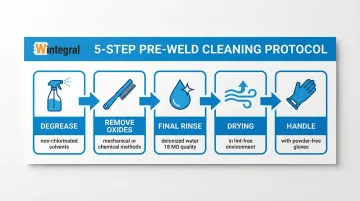
For critical applications, welding occurs in Class 100 (ISO Class 5) cleanrooms where particulate counts are strictly controlled.
Shielding Gas Purity
Standard industrial argon isn't pure enough for medical welding. High-purity specifications include:
| Parameter | Specification |
|---|---|
| Purity | ≥99.999% (Grade 5.0) |
| Oxygen | ≤2 ppm |
| Moisture | ≤3 ppm |
| Nitrogen | ≤5 ppm |
Beyond torch shielding, back purging removes atmospheric gases from the weld root. For stainless steel, purge until oxygen levels drop below 50 ppm.
For titanium, continue purging until oxygen is below 10 ppm. This can take several minutes depending on joint geometry.
Welder Qualification and Certification
While FDA doesn't mandate specific certifications, industry best practice requires welders qualified to AWS B2.1 or ASME Section IX. Qualifications must be specific to the process (GTAW), material (base metal and filler), and joint configuration being welded.
Welders must requalify periodically (typically annually) or whenever they haven't performed a specific process for six months. This ensures skills remain current and consistent with validated procedures.
Choosing a Medical Welding Service Provider
Key Capabilities to Look For
Medical device manufacturers seeking welding partners should verify:
Process Expertise: Experience with multiple welding methods (TIG, laser, micro-plasma) allows providers to recommend the optimal process for each application.
Look for demonstrated experience with medical-grade materials like 316L stainless steel and titanium alloys.
Quality Certifications: ISO 9001:2015 Certified demonstrates commitment to medical device quality management. AWS certification for welding procedures and personnel indicates technical competence.
Material Handling: Proper material storage, handling procedures, and traceability systems ensure materials aren't contaminated or mixed before welding.
Documentation and Traceability
Comprehensive weld records must include:
- Material certifications and heat numbers
- Welding procedure specifications (WPS)
- Welder qualifications and identification
- Process parameters for each weld (current, voltage, travel speed, gas flow)
- Inspection results with acceptance criteria
- Nonconformance documentation and corrective actions
This documentation satisfies regulatory requirements and enables rapid investigation if quality issues arise.
Quality Assurance Processes
Strong welding partners maintain:
- In-house inspection capabilities (visual, dye penetrant, radiographic)
- Statistical process control monitoring critical parameters
- Continuous improvement programs addressing defects and process optimization
- Documented corrective and preventive action (CAPA) systems
Precision welding specialists offering multiple processes (including TIG, laser, and micro-plasma) can match the right technology to each application. Providers handling diverse component sizes with consistently low defect rates demonstrate the precision and quality control medical applications demand.
Frequently Asked Questions
What is a medical welder?
A medical welder is a certified professional who joins metals for medical devices while adhering to strict regulatory standards. They must understand biocompatibility requirements, contamination control, and documentation protocols specific to medical applications.
What makes TIG welding better than other methods for medical devices?
TIG welding produces contamination-free welds with no flux residue or spatter, making it ideal for biocompatible applications. The precise heat control prevents distortion in thin-walled components while simplifying regulatory compliance.
What certifications are required for medical welding?
Key certifications include AWS welder qualifications (per AWS B2.1), ISO 9001:2015 Certified for quality management, and FDA Quality System Regulation (21 CFR Part 820) adherence. Specific applications may require ASME Section IX or ASME BPE compliance.
Can all metals used in medical devices be TIG welded?
Most medical-grade metals—including stainless steel 316L/304, titanium alloys, and commercially pure titanium—can be TIG welded with appropriate parameters. Nitinol requires specialized techniques due to heat sensitivity. Each material requires validated procedures specific to its characteristics.
How is weld quality verified in medical device manufacturing?
Quality verification combines visual inspection per ASME BPE or AWS D18.1 standards, dye penetrant testing for surface defects, and radiographic examination for internal defects. Qualification welds undergo destructive testing to validate procedures before production.
What is the difference between medical-grade and industrial TIG welding?
Medical-grade welding requires higher purity shielding gases (≥99.999% argon vs. 99.9% for industrial), complete documentation and traceability, validated processes, and stricter contamination control. Every aspect is documented to satisfy FDA and ISO standards requirements.


